SGGPO
21,000 vials of injectable Phenobarbital have just arrived in Vietnam after a long interruption, promptly supplementing an important source of medicine for the treatment of severe hand, foot and mouth disease in Ho Chi Minh City.
 |
On August 9, the Ho Chi Minh City Department of Health announced that 21,000 vials of injectable Phenobarbital had just arrived in Vietnam after a long interruption, timely supplementing the drug supply for the treatment of severe hand, foot and mouth disease (HFMD) at 3 specialized pediatric hospitals and the Tropical Diseases Hospital in the city. Previously, the Ho Chi Minh City Department of Health also received 1,000 vials of Gamma-globulin for the treatment of HFMD.
According to the Ho Chi Minh City Department of Health, at the end of 2020, the domestic supply of Phenobarbital was limited and interrupted for a long time. The Department's professional council met and agreed to choose other temporary sedatives currently on the market such as Diazepam, Midazolam... while waiting for Vietnamese pharmaceutical companies to find sources of supply from other countries. At the same time, the Department of Health also sent a dispatch requesting the Drug Administration of Vietnam, the Ministry of Health to support in finding sources of supply of drugs and importing companies to meet the needs of Phenobarbital treatment of hospitals.
On June 22, the Drug Administration of Vietnam responded that the manufacturer Daihan Pharm Co., Ltd. would no longer produce Danotan. The Administration has sent a document requesting drug import companies to contact foreign partners to find alternative drug sources.
By February 1, 2023, Central Pharmaceutical Joint Stock Company CPC1 had found a replacement supplier of Phenobarbital and was granted a license by the Drug Administration of Vietnam to import 21,000 vials of Barbit injection 200mg/ml (phenobarbital injection) from the manufacturer Incepta Pharmaceutical Ltd (Bangladesh) to Vietnam.
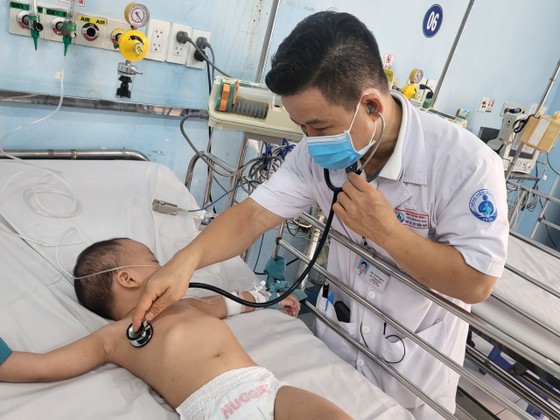 |
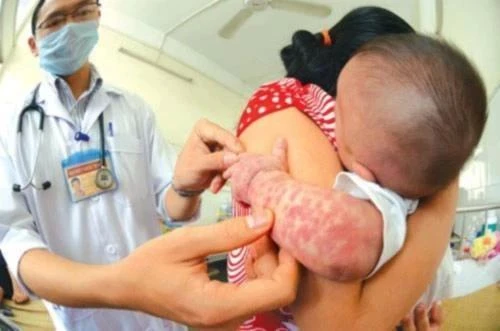
Doctors at Children's Hospital 1 are examining a child with severe hand, foot and mouth disease. |
By July 31, after a long period of paperwork (because Phenobarbital is a psychotropic drug in the group of drugs that must be specially controlled, requiring an export license from the competent authority of the exporting country), 21,000 tubes of injectable Phenobarbital arrived in Vietnam and were immediately supplied by Central Pharmaceutical Joint Stock Company CPC1 to specialized pediatric hospitals and the Ho Chi Minh City Hospital for Tropical Diseases to serve the treatment of TCM diseases at the peak of the epidemic.
Phenobarbital is an important treatment drug included in the TCM treatment regimen issued by the Ministry of Health in Decision No. 1003/QD-BYT dated March 30, 2012. According to this regimen, Phenobarbital can be taken orally or by injection, with injection being preferred when the patient is seriously ill. Phenobarbital is used for children with many advantages, few side effects, and has been used by doctors for pediatric patients for a long time.
In TCM, Phenobarbital plays a role in stopping prolonged seizures and preventing seizure recurrence. In addition, this drug also has a sedative effect, reducing neurological manifestations and startles in children. In addition, when there is cerebral edema (in severe cases), the drug helps reduce cerebral edema and reduce the brain's need for oxygen consumption.
Injectable phenobarbital has a rapid onset of action in a short period of time (5 minutes after injection), reaches peak effect quickly (15-30 minutes) and has a relatively short duration of action (6 hours). Compared to the oral form, the onset of action is delayed (> 60 minutes), the duration of action is long (10-12 hours) and depends on the absorption of each child. Injectable phenobarbital is indicated for children with TCM in the risk group and severe group (group 2b, group 3).
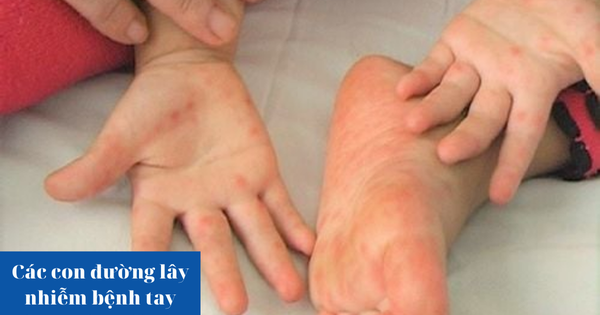
According to statistics from the Ho Chi Minh City Department of Health, from the beginning of 2023 to now, Ho Chi Minh City has had more than 15,753 cases of TCM. Every day, there are hundreds of new cases and many severe and critical cases transferred from other localities. Of which, most of the hospitalized cases are children under 6 years old.
To proactively supply domestic resources, the Drug Administration has granted a license to import Phenobarbital raw materials to Danapha Pharmaceutical Joint Stock Company to produce injectable Phenobarbital. This company is implementing the production plan.
Source




![[Photo] General Secretary To Lam receives Japanese Ambassador to Vietnam Ito Naoki](https://vstatic.vietnam.vn/vietnam/resource/IMAGE/2025/4/3/3a5d233bc09d4928ac9bfed97674be98)

![[Photo] Moment of love: Myanmar people are moved to thank Vietnamese soldiers](https://vstatic.vietnam.vn/vietnam/resource/IMAGE/2025/4/3/9b2e07196eb14aa5aacb1bc9e067ae6f)
![[Photo] Special relics at the Vietnam Military History Museum associated with the heroic April 30th](https://vstatic.vietnam.vn/vietnam/resource/IMAGE/2025/4/3/a49d65b17b804e398de42bc2caba8368)



















































































Comment (0)